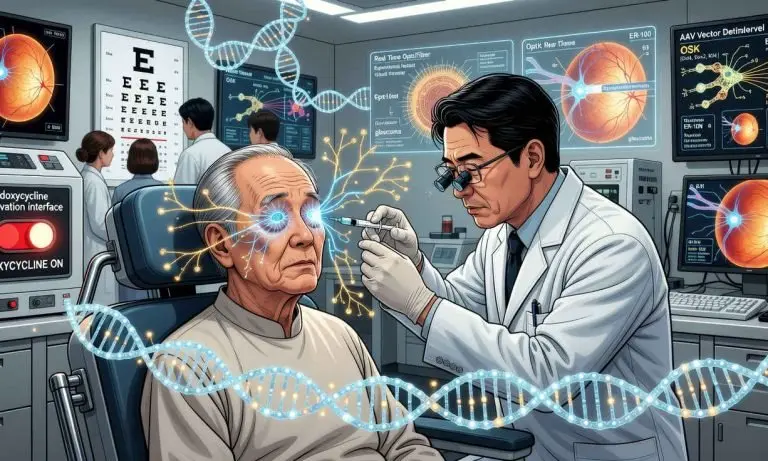
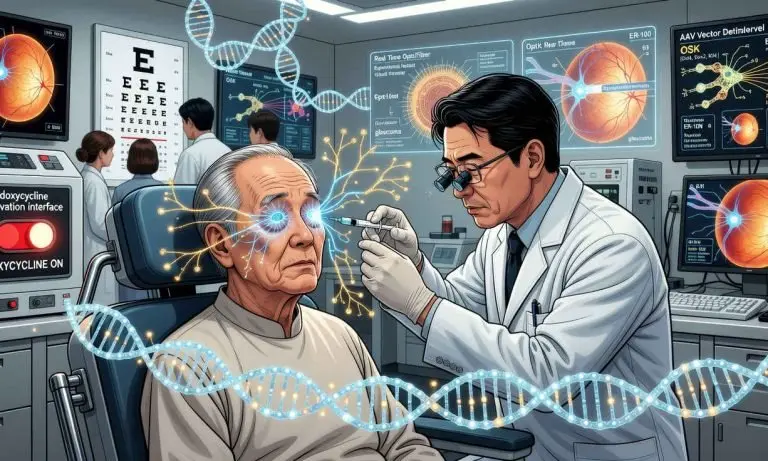
JUST IN: Life Biosciences has initiated the first human clinical trial of a therapy based on partial epigenetic reprogramming, an approach that seeks to restore the function of aging cells without erasing their identity.
The candidate, ER 100, has been approved by the FDA and is already being tested in patients with open angle glaucoma and NAION (non invasive glaucoma).
The FDA has authorized Life Biosciences to begin a Phase 1 human trial of ER 100, a partial epigenetic reprogramming therapy.
The candidate, ER 100, has been approved by the FDA and is already being tested in patients with open angle glaucoma and NAION (non invasive glaucoma).
The FDA has authorized Life Biosciences to begin a Phase 1 human trial of ER 100, a partial epigenetic reprogramming therapy.